A Rare Case of Cutaneous Actinomadura geliboluensis Infection: A Case Report and Review of the Literature
Georgia Kliampa*, Theodora Douvali, Eleni Paparizou, Asimina Papadopoulou, Vasiliki Chasapi
Andreas Syggros Hospital of Athens, Greece
*Corresponding author: Georgia Kliampa, Andreas Syggros Hospital of Athens, Greece.
Received: 07 March 2026; Accepted: 13 March 2026; Published: 08 April 2026
Article Information
Citation: Georgia Kliampa, Theodora Douvali, Eleni Paparizou, Asimina Papadopoulou, Vasiliki Chasapi. A Rare Case of Cutaneous Actinomadura geliboluensis Infection: A Case Report and Review of the Literature. Archives of Microbiology and Immunology. 10 (2026): 42-46.
View / Download Pdf Share at FacebookAbstract
Actinomadura geliboluensis is an uncommon actinomycete species with extremely limited documentation in human disease. An exceptionally rare case of actinomycetoma caused by Actinomadura geliboluensis was identified in the “Venereology and Dermatology Hospital of Athens, A. Syggros” in a 45-year-old female patient presented with acute-onset erythematous nodules on the dorsal surface of the right hand and lymphatic spread along the forearm. Initial outpatient treatment was unsuccessful. Biopsy and tissue cultures were performed, revealing Actinomadura geliboluensis, an actinomycete of the genus Actinomadura. The patient was treated with intravenous antibiotics, followed by long-term oral therapy, resulting in complete remission after six months. This case emphasizes that novel actinomycetes strains can cause mycetoma even in non-endemic areas, independent of travel or immunosuppressive conditions. Diagnostic consideration and early diagnosis are crucial for appropriate therapeutic management and preventing serious complications.
Keywords
Actinomadura geliboluensis, rare actinomycetes, actinomycetoma, Europe, diagnostic consideration, drug susceptibility
Article Details
Introduction
Mycetoma is an uncommon, chronic granulomatous disease of the skin and subcutaneous tissue, caused by fungi or bacteria. It is also called “Madura foot” due to Gill's first report of the disease in the Indian town of Madura (1842). In 1860, Carter introduced the name mycetoma, describing its fungal etiology. In 1913, Pinoy described the mycetoma produced by aerobic bacteria that belong to the actinomycete group and classified mycetomas as eymycetoma (due to true fungi) and actinomycetoma (due to aerobic bacteria)[1,2]. Mycetoma’s clinical picture, whether caused by fungi or bacteria, is quite similar. Microorganisms usually are introduced as a result of a minor trauma or injury. Mycetoma mainly affects men and is frequently linked to certain occupations. It is commonly seen in farmers, field workers, landscapers, herders, and others who regularly come into contact with soil as part of their work[3].
Painless firm swellings, nodules, abscess formation, and fistulae characterize mycetoma. The fistulae drain serous or purulent material, which contains grains, consisting of colonies of the causative agent [4,5]. It can expand through the tissues and can lead to the destruction of muscles and adjacent bones. Lymphatic spread of mycetoma occurs only in 1-3% of affected patients and this is more frequent in actinomycetoma [3,4]. Mycetoma due to actinomycetes should be distinguished from actinomycosis, an endogenous suppurative infection brought on by Actinomyces species, such Actinomyces israelii, that typically affects the cervicofacial, thoracic, and pelvic sites. Branching bacteria that are non-acid-fast anaerobic or microaerophilic, cause actinomycosis, while the actinomycetes that cause actinomycetoma are always aerobic and are sometimes weakly acid-fast [6].
Mycetoma was recognized as a neglected tropical disease (NTD), by the World Health Organization in 2016, due to inadequate global surveillance and the absence of control programs [5,7,8,9,10,11,12]. It is often misdiagnosed and mismanaged. It is endemic in the so-called ‘mycetoma belt’, between 15° latitude south and 30° latitude north. Sporadic cases have been reported also in countries with temperate climates, specifically in South-Eastern Europe[7].
Actinomadura geliboluensis belongs to Actinomadura. The genus Actinomadura was proposed by Lechevalier & Lechevalier (1970), and revised descriptions have been provided by Zhang et al. (2001) and Miyadoh & Miyara (2001). Actinomadura geliboluensis was first isolated in 2012 in Canakkale, Turkey from soil samples. It is an aerobic, gram-positive, non-acid-alcohol-fast, non-motile actinomycete[13]. This rare actinomycetes strain was isolated from culture tissue and it is the first report of Actinomadura geliboluensis being isolated from the skin and causing mycetoma in a country outside the mycetoma belt.
Case Presentation
A 45-year-old female patient presented to our hospital with soft, erythematous nodules on the dorsal surface of the right hand. The patient did not have fever or other systematic symptoms. She reported a subungual injury while nail clipping two months ago. She owned several pets (dog, canary, rabbit, fish) and had no recent travel history. Past medical history included thyroid cancer surgery six years earlier. According to the patient, a few days after the subungual injury, she developed redness with a linear pattern from the right index finger to the right axilla as well as painful, swollen lymph nodes in the right axilla. After a few days, the soft, erythematous nodules appeared on the dorsal surface of the right hand. She visited a dermatologist and received oral antibiotic treatment (ciprofloxacin and clindamycin) for 22 days, with no significant improvement.
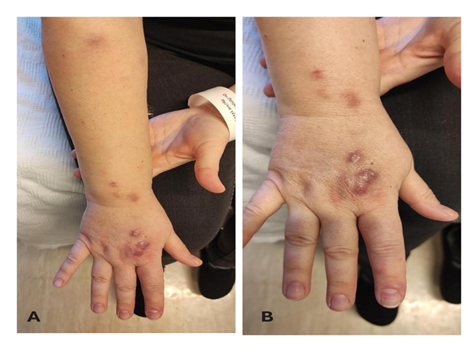
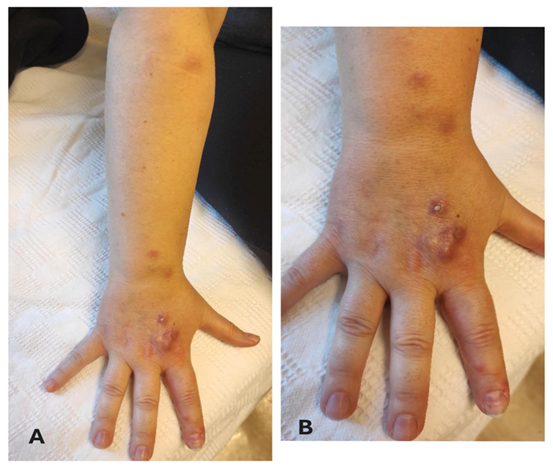
At admission to the hospital, the clinical examination revealed soft erythematous nodules on an erythematous base, painful to the touch, with a lymphatic distribution up to the upper forearm, without swollen lymph nodes (Figure 1A,1B). Laboratory tests were normal. A skin punch biopsy and tissue culture were performed on one of the nodules.
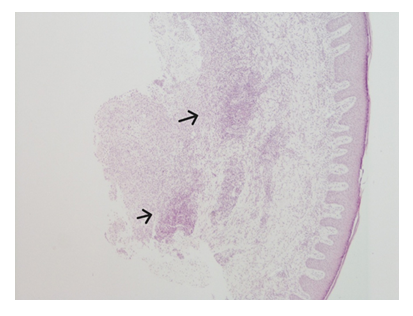
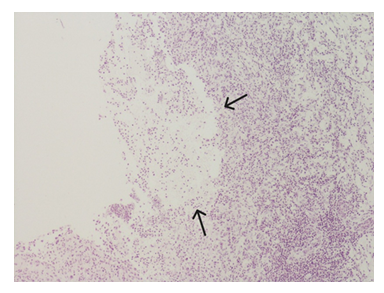
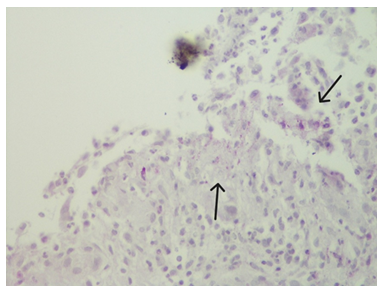
Histological examination revealed that the skin fragment had dense, intense mixed-type inflammation throughout the entire thickness of the dermis (Figure2), partially abscess-forming (Figure 3). PAS staining showed the presence of granules intracellularly and extracellularly (Figure 4), and Actinomadura geliboluensis was isolated from tissue culture, using the Maldi-Tof method.
A: Lymphatic distribution up to the upper forearm.
B: Erythematous nodules on the dorsal surface of the right hand.
The black arrow indicates the area of dense, intense mixed-type inflammatory infiltrate extending throughout the full thickness of the dermis.
The black arrow indicates the abscess.
The black arrow indicates the granules.
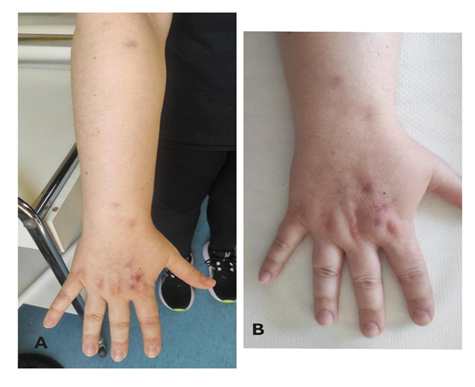
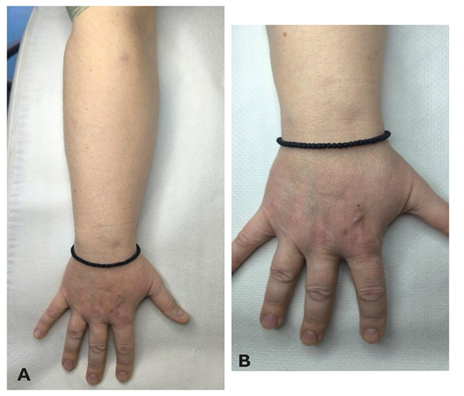
A chest X-ray and pulmonary evaluation were performed with no signs of pulmonary infection. A hand X-ray excluded the spread of the infection. Intravenous antibiotic therapy was initiated with piperacillin-tazobactam (4.5 g IV q8hr). After three days of treatment the patient experienced clinical improvement with reduced swelling of the nodules. Intravenous piperacillin-tazobactam was administered for 11 days, leading to further clinical response with a gradual reduction in the size of the nodules and erythema (Figure 5A, 5B). The patient remained afebrile throughout the hospitalization, and no signs of infection spread to other organs were observed. She was discharged with instructions to continue oral antibiotic therapy with doxycycline. After six weeks of treatment, the patient demonstrated significant improvement, and physical examination revealed smaller, erythematous nodules. At three months follow-up, nodules were present only on the dorsal surface of the right forearm, while the right hand showed no nodules, just post-inflammatory hyperpigmentation (Figure 6A, 6B). Laboratory tests remained normal, and the same treatment continued. After five months of doxycycline therapy, all nodules resolved, leaving post-inflammatory hyperpigmentation. The patient continued doxycycline at 100 mg daily for an additional month, completing a total of six months of treatment with complete clinical remission (Figure 7A, 7B).
A, B: Reduction in the size of the nodules
- Nodules on the forearm.
- Post-inflammatory hyperpigmentation on the right hand.
Discussion
This case is notable for highlighting mycetoma caused by an extremely rare actinomycete species in a non-endemic area. Mycetoma is a rare disease, endemic in many tropical and subtropical areas. It can progress devastatingly and lead to amputation by spreading to adjacent organs[13,7]. High clinical suspicion is necessary to prevent severe complications. Mycetoma can also occur in countries with temperate climates, outside the mycetoma belt, where most physicians are unfamiliar with this disease. Recent increases in immigration and worldwide travel, as well as good climate conditions for the survival of the causative agents, may be the cause of this [3,7,9]. Male rural workers are more likely to develop mycetoma, which typically affects anatomical areas like the feet and hands. Clinically, it begins as painless subcutaneous lumps and develops into many inflamed sinuses with grain-containing purulent discharge over time[3-5]. In our patient’s case no draining sinus tracts were observed, but numerous subcutaneous masses along her right upper arm. Given the often painless nature of early symptoms and the rarity of mycetoma, diagnosis may be delayed[3].
Mycetoma can be diagnosed by direct examination of grains from the discharge of the sinuses. In cases where no sinus tracts are present, like our case, a fine-needle aspiration or punch biopsy must be performed. Grains vary in size, color, and texture and are characteristic of the causative agent. For establishing the diagnosis biopsy and microbiological cultures are fundamental. For precise bacterial or fungal genus and species identification, molecular methods such as 16S rRNA gene sequencing, or matrix-assisted laser desorption ionization-time-of-flight mass spectrometry (MALDI-TOF MS) are essentiall [1,2,4]. Imaging diagnostics including X-ray imaging, ultrasound (US), computer tomography (CT), and magnetic resonance imaging (MRI) allow assessment of early diagnosis and expansion of mycetoma. The so-called dot-in-circle sign in MRI is a highly specific sign for mycetoma [1,10,11].
Mycetoma is treated with a combination of surgical excision and long-term pharmacotherapy. Surgery is recommended for a localized mycetoma that can be excised completely and is infrequently indicated in actinomycetoma. A wide range of treatment options are available for actinomycetomas, as they respond to antibiotic treatment. Trimethoprim-sulfamethoxazole, dapsone and streptomycin sulfate or amicasin have been used. Trimethoprim-sulfomethoxazole (7,5-40mg/kg/day) twice daily is a standard therapy for actinomycetomas over months. Amicasin(15mg/kg/day) should be added in certain anatomical sites (eg thorax, head) and persistent cases, the so-called Welsh regimen. Adding rifampin to the Welsh regimen (modified Welsh regimen) is used to avoid recurrence. Other antimicrobials such as amoxicillin-clavulanic acid, imipenem, meropenem, doxycycline and linezolid have been used with variable success. Itraconazole is the main pharmacotherapeutic option for treating eumycetoma [2,8,12].
Regarding the therapeutic management of our case, we relied on international literature and especially on the single reported case of infection by Actinomadura geliboluensis, a case of pneumonia by Actinomadura geliboluensis treated with piperacillin-tazobactam. The results of this case showed that Actinomadura geliboluensis was susceptible to tetracyclines, quinolones and sulfonamides and resistant to carbapenems, penicillins and cephalosporins. It also appeared that Actinomadura geliboluensis was sensitive to piperacillin-tazobactam[14]. Considering these results, our patient received piperacillin-tazobactam intravenously during her hospitalization and doxycycline as an outpatient.
Conclusions
This case highlights the first known instance of mycetoma caused by Actinomadura geliboluensis, a rare actinomycete strain. Previously reported only in a case of pneumonia, this bacterium presented in our patient following minor subungual trauma, leading to multiple erythematous nodules in a lymphatic distribution. Our findings indicate that uncommon pathogens such as Actinomadura geliboluensis can cause mycetoma even outside the mycetoma belt, without clear associations to travel or immunosuppressive conditions. This case underscores the importance of considering rare organisms in persistent cutaneous infections and highlights the need for individualized, culture-guided therapy. Since infections caused by Actinomadura geliboluensis are rare, more research is needed to understand its behavior, resistance to antibiotics, and best treatment options. Greater awareness and early detection of this emerging pathogen are vital to improving outcomes for patients with skin or systemic infections.
References
- Nenoff P, Van De Sande WWJ, Fahal AH, et al. Eumycetoma and actinomycetoma--an update on causative agents, epidemiology, pathogenesis, diagnostics and therapy. J Eur Acad Dermatol Venereol 29 (2015): 1873-83.
- Reis CMS, Reis-Filho EG de M. Mycetomas: an epidemiological, etiological, clinical, laboratory and therapeutic review. An Bras Dermatol 93 (2018): 8-18.
- Fahal AH, Suliman SH, Hay R. Mycetoma: The Spectrum of Clinical Presentation. Trop Med Infect Dis (2018).
- Venkatswami S, Sankarasubramanian A, Subramanyam S. The madura foot: looking deep. Int J Low Extrem Wounds 11 (2012): 31-42.
- de la Garza JAC, Welsh O, Cuéllar-Barboza A, et al. Clinical characteristics and treatment of actinomycetoma in northeast Mexico: A case series. PLoS Negl Trop Dis (2020).
- Najmi AH, Najmi IH, Tawhari MMH, et al. Cutaneous actinomycosis and long-term management through using oral and topical antibiotics: A case report. Clin Pract 8 (2018): 1102.
- Buonfrate D, Gobbi F, Angheben A, et al. Autochthonous cases of mycetoma in Europe: report of two cases and review of literature PLoS One (2014).
- Verma P, Jha A. Mycetoma: reviewing a neglected disease. Clin Exp Dermatol 44 (2019): 123-9.
- van de Sande WWJ. Global burden of human mycetoma: a systematic review and meta-analysis. PLoS Negl Trop Dis 7 (2013).
- Aggarwal A, Gupta S, Chopra D, et al. “Dot-in-circle sign:” An ultrasound and magnetic resonance imaging sign for mycetoma. Medicine India 1 (2022): 10.
- Basirat A, Boothe E, Mazal AT, et al. Soft tissue mycetoma: “Dot-in-circle” sign on magnetic resonance imaging. Radiol Case Rep 15 (2020): 467-73.
- Welsh O, Vera-Cabrera L, Welsh E, et al. Actinomycetoma and advances in its treatment. Clin Dermatol 30 (2012): 372-81.
- Sazak A, Camas M, Spröer C, et al. Actinomadura geliboluensis sp. nov., isolated from soil. Int J Syst Evol Microbiol 62 (2012): 2011-7.
- Yu Y, Yang G, Wang Y, et al. A Rare Strain Actinomadura geliboluensis Was First Isolated from the Bronchoalveolar Lavage Fluid of a Patient with Pneumonia. Infect Drug Resist 16 (2023): 3101-8.


 Impact Factor:
* 3.5
Impact Factor:
* 3.5
 Acceptance Rate:
71.36%
Acceptance Rate:
71.36%
 Time to first decision: 10.4 days
Time to first decision: 10.4 days
 Time from article received to acceptance:
2-3 weeks
Time from article received to acceptance:
2-3 weeks

